Water is the only substance that naturally exists in 3 forms, as a solid (ice), as a liquid (the water we drink) and as a gas (water vapor). Water in the atmosphere exists in all three forms with the vast majority of water being in the vapor form.
Visible water in the air
Clouds are the visible form of liquid water in the air. Clouds are formed when water vapor in the air condenses into liquid droplets that are so small that they are light enough to float in the air. For this change (condensation) in form to occur, water needs a non-gaseous surface. Physical particles that serve this purpose are known as cloud condensation nuclei (CCN).
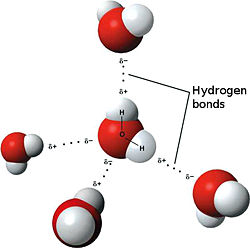
For clouds to be seen, water droplets need to coalesce (combine) into larger droplets. This coalescence typically happens when individual droplets collide as they move through the air. Water droplets combine with some difficulty (a direct result of the hydrogen bonds discussed in my post dated December 4, 2010) as illustrated by the gravity-fueled "dance" of a drop of liquid water that is falling onto a larger liquid water body, in the video below:
For clouds to produce rain drops, the CCN-created water droplets need to grow about a million times larger through numerous collisions.
Invisible (to the human eye) liquid water in the Air
Liquid water droplets that are smaller than 0.02 mm in size that find a physical particle suitable for formation (from water vapor), can also be trapped between particles and stay in that 'normal' state for extended periods of time. Aerosol particles, by holding water droplets captive, contribute a continued and longer-term worsening impact on global warming.
Initiatives that are efficient and effective at extracting this "trapped" water from the atmosphere and inhibiting continuing entrapment would produce both fresh drinking water and reduce Global Warming.
References:
http://www.atmosphere.mpg.de/enid/850ac2615b35b9f50abbc96fa4273dbb,0/1__Clouds/-_Water_in_the_atmosphere_25n.html
http://www.youtube.com/watch?v=6KKNnjFpGto&feature=BF&playnext=1&list=QL